
Cerium(IV) oxide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cerium dioxide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
For the other compound also known as cerium oxide, see Cerium(III) oxide.
Cerium(IV) oxide, also known as ceric oxide, ceric dioxide, ceria, cerium oxide or cerium dioxide, is an oxide of the rare-earth metal cerium. It is a pale yellow-white powder with the chemical formula CeO2. It is an important commercial product and an intermediate in the purification of the element from the ores. The distinctive property of this material is its reversible conversion to a non-stoichiometric oxide.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Cerium(IV) oxide | |
| Other names
Ceric oxide, Ceria, Cerium dioxide | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.013.774 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| CeO2 | |
| Molar mass | 172.115 g/mol |
| Appearance | white or pale yellow solid, slightly hygroscopic |
| Density | 7.215 g/cm3 |
| Melting point | 2,400 °C (4,350 °F; 2,670 K) |
| Boiling point | 3,500 °C (6,330 °F; 3,770 K) |
| insoluble | |
| +26.0·10−6 cm3/mol | |
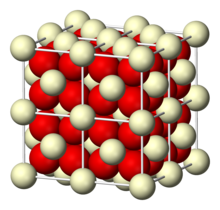
| Structure | |
| cubic crystal system, cF12 (fluorite)[1] | |
| Fm3m, #225 | |
α = 90°, β = 90°, γ = 90° | |
| Ce, 8, cubic O, 4, tetrahedral | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds |
Cerium(III) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
