Nitrosyl bromide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Nitrosyl bromide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
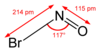
Nitrosyl bromide is the chemical compound with the chemical formula NOBr. It is a red gas with a condensing point just below room temperature.[1] It reacts with water.[1]
Quick Facts Identifiers, Properties ...
 | |||
| |||
| Identifiers | |||
|---|---|---|---|
3D model (JSmol) |
|||
| ChemSpider |
| ||
PubChem CID |
|||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| NOBr | |||
| Molar mass | 109.910 g/mol | ||
| Appearance | Red gas | ||
| Boiling point | 14.5 °C (58.1 °F; 287.6 K) | ||
Refractive index (nD) |
1.524 | ||
| Related compounds | |||
Related compounds |
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
Nitrosyl bromide can be formed by the reversible reaction of nitric oxide with bromine.[2] This reaction is of interest as it is one of very few third-order homogeneous gas reactions. NOBr is prone to photodissociation at standard pressure and temperature.
- 2 NO + Br2 ⇌ 2 NOBr
Another way to make it is by way of nitric oxide reacting with potassium bromide.[1]
- 2NO2 + KBr → BrNO + KNO3