
Potassium carbonate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Potassium carbonate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
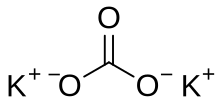
Potassium carbonate is the inorganic compound with the formula K2CO3. It is a white salt, which is soluble in water and forms a strongly alkaline solution. It is deliquescent, often appearing as a damp or wet solid. Potassium carbonate is mainly used in the production of soap and glass.[3] Commonly, it can be found as the result of leakage of alkaline batteries.[4]
Quick Facts Names, Identifiers ...
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Potassium carbonate | |
| Other names
Carbonate of potash, dipotassium carbonate, sub-carbonate of potash, pearl ash, potash, salt of tartar, salt of wormwood. | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.665 |
| E number | E501(i) (acidity regulators, ...) |
PubChem CID |
|
| RTECS number |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| K 2CO 3 | |
| Molar mass | 138.205 g/mol |
| Appearance | White, hygroscopic solid |
| Density | 2.43 g/cm3 |
| Melting point | 891 °C (1,636 °F; 1,164 K) |
| Boiling point | Decomposes |
| 110.3 g/100 mL (20 °C) 149.2 g/100 mL (100 °C) | |
| Solubility | |
| Acidity (pKa) | 10.25 |
| −59.0·10−6 cm3/mol | |
| Thermochemistry[1] | |
Heat capacity (C) |
114.4 J·mol−1·K−1 |
Std molar entropy (S⦵298) |
155.5 J·mol−1·K−1 |
Std enthalpy of formation (ΔfH⦵298) |
−1151.0 kJ·mol−1 |
Gibbs free energy (ΔfG⦵) |
−1063.5 kJ·mol−1 |
Enthalpy of fusion (ΔfH⦵fus) |
27.6 kJ·mol−1 |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H302, H315, H319, H335 | |
| P261, P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
1870 mg/kg (oral, rat)[2] |
| Safety data sheet (SDS) | ICSC 1588 |
| Related compounds | |
Other anions |
Potassium bicarbonate |
Other cations |
Lithium carbonate Sodium carbonate Rubidium carbonate Caesium carbonate |
Related compounds |
Ammonium carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
