
Selenocysteine
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Selenocysteine?
Summarize this article for a 10 year old
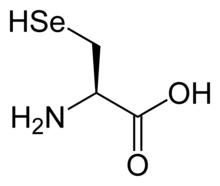
Selenocysteine (symbol Sec or U,[4] in older publications also as Se-Cys)[5] is the 21st proteinogenic amino acid. Selenoproteins contain selenocysteine residues. Selenocysteine is an analogue of the more common cysteine with selenium in place of the sulfur.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Selenocysteine | |
| Systematic IUPAC name
3-Selanyl-L-alanine (semisystematic name)
2-Amino-3-selanylpropanoic acid (fully systematic name) | |
| Other names
L-Selenocysteine; Selenium-cysteine | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| DrugBank |
|
| ECHA InfoCard | 100.236.386 |
| KEGG |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H7NO2Se | |
| Molar mass | 168.065 g·mol−1 |
| Properties | |
| Acidity (pKa) | 5.24,[2] 5.43[3] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Selenocysteine is present in several enzymes (for example glutathione peroxidases, tetraiodothyronine 5′ deiodinases, thioredoxin reductases, formate dehydrogenases, glycine reductases, selenophosphate synthetase 2, methionine-R-sulfoxide reductase B1 (SEPX1), and some hydrogenases). It occurs in all three domains of life, including important enzymes (listed above) present in humans.[6]
Selenocysteine was discovered in 1974[7] by biochemist Thressa Stadtman at the National Institutes of Health.[8]