
Sulfur hexafluoride
Chemical compound and greenhouse gas / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sulfur hexafluoride?
Summarize this article for a 10 year old
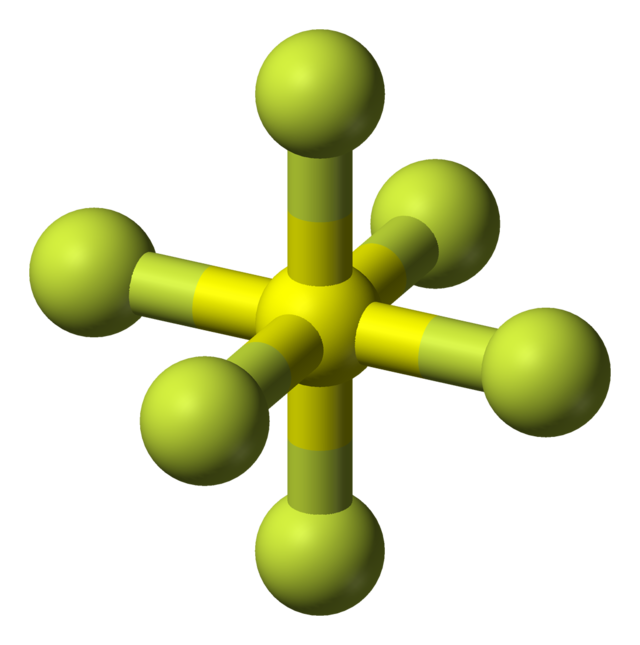
Sulfur hexafluoride or sulphur hexafluoride (British spelling) is an inorganic compound with the formula SF6. It is a colorless, odorless, non-flammable, and non-toxic gas. SF
6 has an octahedral geometry, consisting of six fluorine atoms attached to a central sulfur atom. It is a hypervalent molecule.
| |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Sulfur hexafluoride | |||
| Systematic IUPAC name
Hexafluoro-λ6-sulfane[1] | |||
| Other names
Elagas Esaflon | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.018.050 | ||
| EC Number |
| ||
| 2752 | |||
| KEGG |
| ||
| MeSH | Sulfur+hexafluoride | ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1080 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| SF6 | |||
| Molar mass | 146.05 g·mol−1 | ||
| Appearance | Colorless gas | ||
| Odor | odorless[2] | ||
| Density | 6.17 g/L | ||
| Melting point | −64 °C; −83 °F; 209 K | ||
| Boiling point | −50.8 °C (−59.4 °F; 222.3 K) | ||
| Critical point (T, P) | 45.51±0.1 °C, 3.749±0.01 MPa[3] | ||
| 0.003% (25 °C)[2] | |||
| Solubility | slightly soluble in water, very soluble in ethanol, hexane, benzene | ||
| Vapor pressure | 2.9 MPa (at 21.1 °C) | ||
| −44.0×10−6 cm3/mol | |||
| Thermal conductivity |
| ||
| Viscosity | 15.23 μPa·s[5] | ||
| Structure | |||
| Orthorhombic, oP28 | |||
| Oh | |||
| Orthogonal hexagonal | |||
| Octahedral | |||
| 0 D | |||
| Thermochemistry | |||
Heat capacity (C) |
0.097 kJ/(mol·K) (constant pressure) | ||
Std molar entropy (S⦵298) |
292 J·mol−1·K−1[6] | ||
Std enthalpy of formation (ΔfH⦵298) |
−1209 kJ·mol−1[6] | ||
| Pharmacology | |||
| V08DA05 (WHO) | |||
| License data | |||
| Hazards | |||
| GHS labelling:[7] | |||
 | |||
| Warning | |||
| H280 | |||
| P403 | |||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 1000 ppm (6000 mg/m3)[2] | ||
REL (Recommended) |
TWA 1000 ppm (6000 mg/m3)[2] | ||
IDLH (Immediate danger) |
N.D.[2] | ||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Related sulfur fluorides |
Disulfur decafluoride | ||
Related compounds |
Selenium hexafluoride Sulfuryl fluoride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Typical for a nonpolar gas, SF
6 is poorly soluble in water but quite soluble in nonpolar organic solvents. It has a density of 6.12 g/L at sea level conditions, considerably higher than the density of air (1.225 g/L). It is generally transported as a liquefied compressed gas.
SF
6 has 23,500 times greater global warming potential (GWP) than CO2 as a greenhouse gas (over a 100-year time-frame) but exists in relatively minor concentrations in the atmosphere. Its concentration in Earth's troposphere reached 11.50 parts per trillion (ppt) in October 2023, rising at 0.37 ppt/year.[8] The increase since 1980 is driven in large part by the expanding electric power sector, including fugitive emissions from banks of SF
6 gas contained in its medium- and high-voltage switchgear. Uses in magnesium, aluminium, and electronics manufacturing also hastened atmospheric growth.[9]


