
Amylopectin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Amylopectin?
Summarize this article for a 10 year old
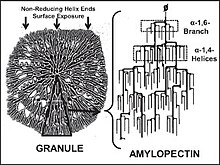
Amylopectin /ˌæmɪloʊˈpɛktɪn/ is a water-insoluble[1][2] polysaccharide and highly branched polymer of α-glucose units found in plants. It is one of the two components of starch, the other being amylose.
 | |
| Identifiers | |
|---|---|
| ChemSpider |
|
| ECHA InfoCard | 100.029.907 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| Properties | |
| [C6H10O5]n | |
| Molar mass | Variable |
| Appearance | White powder |
| Insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Plants store starch within specialized organelles called amyloplasts. To generate energy, the plant hydrolyzes the starch, releasing the glucose subunits. Humans and other animals that eat plant foods also use amylase, an enzyme that assists in breaking down amylopectin, to initiate the hydrolysis of starch.[3]
Starch is made of about 70–80% amylopectin by weight, though it varies depending on the source. For example, it ranges from lower percent content in long-grain rice, amylomaize, and russet potatoes to 100% in glutinous rice, waxy potato starch, and waxy corn. Amylopectin is highly branched, being formed of 2,000 to 200,000 glucose units. Its inner chains are formed of 20–24 glucose subunits.

Dissolved amylopectin starch has a lower tendency of retrogradation (a partial recrystallization after cooking—a part of the staling process) during storage and cooling. For this main reason, the waxy starches are used in different applications mainly as a thickening agent or stabilizer.