Carboxylic acid
Organic compound containing a –C(=O)OH group / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Carboxylic acid?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"COOH" redirects here. For the Bulgarian DJ and producer Ivan Shopov, see Cooh (musician).
Not to be confused with Carbolic acid.
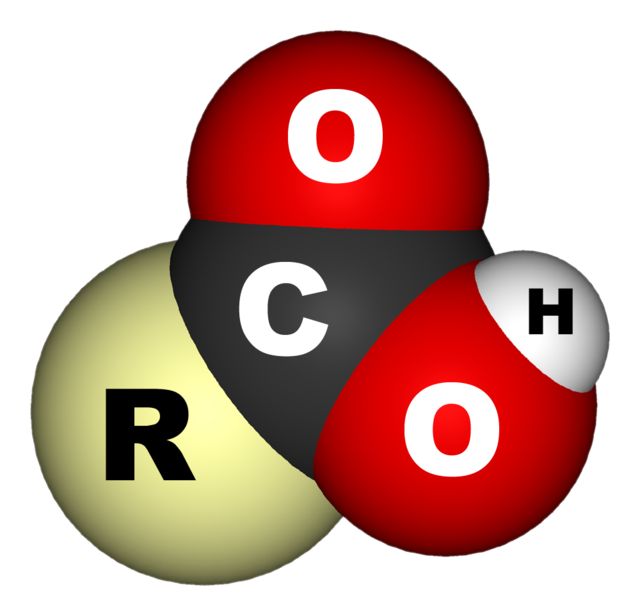
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group (−C(=O)−OH)[1] attached to an R-group. The general formula of a carboxylic acid is often written as R−COOH or R−CO2H, sometimes as R−C(O)OH with R referring to an organyl group (e.g., alkyl, alkenyl, aryl), or hydrogen, or other groups. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion.