
Germane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Germane?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
For a definition of the term "germane", see the Wiktionary entry germane.
Not to be confused with German.
Germane is the chemical compound with the formula GeH4, and the germanium analogue of methane. It is the simplest germanium hydride and one of the most useful compounds of germanium. Like the related compounds silane and methane, germane is tetrahedral. It burns in air to produce GeO2 and water. Germane is a group 14 hydride.
Quick Facts Names, Identifiers ...
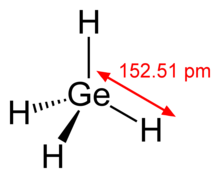 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Germane | |||
| Other names
Germanium tetrahydride Germanomethane Monogermane | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.029.055 | ||
| EC Number |
| ||
| 587 | |||
| KEGG |
| ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2192 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| GeH4 | |||
| Molar mass | 76.62 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Pungent[1] | ||
| Density | 3.3 kg/m3 | ||
| Melting point | −165 °C (−265 °F; 108 K) | ||
| Boiling point | −88 °C (−126 °F; 185 K) | ||
| Low | |||
| Vapor pressure | >1 atm[1] | ||
| Viscosity | 17.21 μPa·s (theoretical estimate)[2] | ||
| Structure | |||
| Tetrahedral | |||
| 0 D | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Toxic, flammable, may ignite spontaneously in air | ||
| GHS labelling: | |||
    | |||
| Danger | |||
| H220, H280, H302, H330 | |||
| P210, P260, P264, P270, P271, P284, P301+P312, P304+P340, P310, P320, P330, P377, P381, P403, P403+P233, P405, P410+P403, P501 | |||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
None[1] | ||
REL (Recommended) |
TWA 0.2 ppm (0.6 mg/m3)[1] | ||
IDLH (Immediate danger) |
N.D.[1] | ||
| Safety data sheet (SDS) | ICSC 1244 | ||
| Related compounds | |||
Related compounds |
Methane Silane Stannane Plumbane Germyl | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close


