Portal:Minerals
Wikipedia portal for content related to Minerals / From Wikipedia, the free encyclopedia
Portal maintenance status: (May 2019)
|
The Minerals Portal

In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.
The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks.
The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases.
Some natural solid substances without a definite crystalline structure, such as opal or obsidian, are more properly called mineraloids. If a chemical compound occurs naturally with different crystal structures, each structure is considered a different mineral species. Thus, for example, quartz and stishovite are two different minerals consisting of the same compound, silicon dioxide. (Full article...)
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...)
Selected articles
- Image 1
A rock containing three crystals of pyrite (FeS2). The crystal structure of pyrite is primitive cubic, and this is reflected in the cubic symmetry of its natural crystal facets.
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of these crystals:- Primitive cubic (abbreviated cP and alternatively called simple cubic)
- Body-centered cubic (abbreviated cI or bcc)
- Face-centered cubic (abbreviated cF or fcc)
Note: the term fcc is often used in synonym for the cubic close-packed or ccp structure occurring in metals. However, fcc stands for a face-centered-cubic Bravais lattice, which is not necessarily close-packed when a motif is set onto the lattice points. E.g. the diamond and the zincblende lattices are fcc but not close-packed.
Each is subdivided into other variants listed below. Although the unit cells in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. (Full article...) - Image 2

Tourmaline (/ˈtʊərməlɪn, -ˌliːn/ TOOR-mə-lin, -leen) is a crystalline silicate mineral group in which boron is compounded with elements such as aluminium, iron, magnesium, sodium, lithium, or potassium. This gemstone comes in a wide variety of colors.
The name is derived from the Sinhalese tōramalli (ටෝරමල්ලි), which refers to the carnelian gemstones. (Full article...) - Image 3A lustrous crystal of zircon perched on a tan matrix of calcite from the Gilgit District of Pakistan
Zircon (/ˈzɜːrkɒn, -kən/) is a mineral belonging to the group of nesosilicates and is a source of the metal zirconium. Its chemical name is zirconium(IV) silicate, and its corresponding chemical formula is ZrSiO4. An empirical formula showing some of the range of substitution in zircon is (Zr1–y, REEy)(SiO4)1–x(OH)4x–y. Zircon precipitates from silicate melts and has relatively high concentrations of high field strength incompatible elements. For example, hafnium is almost always present in quantities ranging from 1 to 4%. The crystal structure of zircon is tetragonal crystal system. The natural color of zircon varies between colorless, yellow-golden, red, brown, blue, and green.
The name derives from the Persian zargun, meaning "gold-hued". This word is changed into "jargoon", a term applied to light-colored zircons. The English word "zircon" is derived from Zirkon, which is the German adaptation of this word. Yellow, orange, and red zircon is also known as "hyacinth", from the flower hyacinthus, whose name is of Ancient Greek origin. (Full article...) - Image 4
Graphite (/ˈɡræfaɪt/) is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3 million metric tons per year in 2022) for uses in pencils, lubricants, and electrodes. Under high pressures and temperatures it converts to diamond. It is a good (but not excellent) conductor of both heat and electricity. (Full article...) - Image 5Quartz crystal cluster from Brazil
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is, therefore, classified structurally as a framework silicate mineral and compositionally as an oxide mineral. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar.
Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at 573 °C (846 K; 1,063 °F). Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold.
There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Europe and Asia.
Quartz is the mineral defining the value of 7 on the Mohs scale of hardness, a qualitative scratch method for determining the hardness of a material to abrasion. (Full article...) - Image 6Malachite from the Democratic Republic of the Congo
Malachite is a copper carbonate hydroxide mineral, with the formula Cu2CO3(OH)2. This opaque, green-banded mineral crystallizes in the monoclinic crystal system, and most often forms botryoidal, fibrous, or stalagmitic masses, in fractures and deep, underground spaces, where the water table and hydrothermal fluids provide the means for chemical precipitation. Individual crystals are rare, but occur as slender to acicular prisms. Pseudomorphs after more tabular or blocky azurite crystals also occur. (Full article...) - Image 7Amethyst cluster from Artigas, Uruguay
Amethyst is a violet variety of quartz. The name comes from the Koine Greek αμέθυστος amethystos from α- a-, "not" and μεθύσκω (Ancient Greek) methysko / μεθώ metho (Modern Greek), "intoxicate", a reference to the belief that the stone protected its owner from drunkenness. Ancient Greeks wore amethyst and carved drinking vessels from it in the belief that it would prevent intoxification.
Amethyst, a semiprecious stone, is often used in jewelry. (Full article...) - Image 8
Beryl (/ˈbɛrəl/ BERR-əl) is a mineral composed of beryllium aluminium silicate with the chemical formula Be3Al2Si6O18. Well-known varieties of beryl include emerald and aquamarine. Naturally occurring hexagonal crystals of beryl can be up to several meters in size, but terminated crystals are relatively rare. Pure beryl is colorless, but it is frequently tinted by impurities; possible colors are green, blue, yellow, pink, and red (the rarest). It is an ore source of beryllium. (Full article...) - Image 9Brazilian trigonal hematite crystal
Hematite (/ˈhiːməˌtaɪt, ˈhɛmə-/), also spelled as haematite, is a common iron oxide compound with the formula, Fe2O3 and is widely found in rocks and soils. Hematite crystals belong to the rhombohedral lattice system which is designated the alpha polymorph of Fe
2O
3. It has the same crystal structure as corundum (Al
2O
3) and ilmenite (FeTiO
3). With this it forms a complete solid solution at temperatures above 950 °C (1,740 °F).
Hematite naturally occurs in black to steel or silver-gray, brown to reddish-brown, or red colors. It is mined as an important ore mineral of iron. It is electrically conductive. Hematite varieties include kidney ore, martite (pseudomorphs after magnetite), iron rose and specularite (specular hematite). While these forms vary, they all have a rust-red streak. Hematite is not only harder than pure iron, but also much more brittle. Maghemite is a polymorph of hematite (γ-Fe
2O
3) with the same chemical formula, but with a spinel structure like magnetite.
Large deposits of hematite are found in banded iron formations. Gray hematite is typically found in places that have still, standing water or mineral hot springs, such as those in Yellowstone National Park in North America. The mineral can precipitate in the water and collect in layers at the bottom of the lake, spring, or other standing water. Hematite can also occur in the absence of water, usually as the result of volcanic activity.
Clay-sized hematite crystals can also occur as a secondary mineral formed by weathering processes in soil, and along with other iron oxides or oxyhydroxides such as goethite, which is responsible for the red color of many tropical, ancient, or otherwise highly weathered soils. (Full article...) - Image 10

Zeolite exhibited in the Estonian Museum of Natural History
Zeolite is a family of several microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula Mn+
1/n(AlO
2)−
(SiO
2)
x・yH
2O where Mn+
1/n is either a metal ion or H+. These positive ions can be exchanged for others in a contacting electrolyte solution. H+
exchanged zeolites are particularly useful as solid acid catalysts.
The term was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material zeolite, from the Greek ζέω (zéō), meaning "to boil" and λίθος (líthos), meaning "stone".
Zeolites occur naturally, but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure that is obtained is examined by the International Zeolite Association Structure Commission (IZA-SC) and receives a three-letter designation. (Full article...) - Image 11Beachy Head is a part of the extensive Southern England Chalk Formation.
Chalk is a soft, white, porous, sedimentary carbonate rock. It is a form of limestone composed of the mineral calcite and originally formed deep under the sea by the compression of microscopic plankton that had settled to the sea floor. Chalk is common throughout Western Europe, where deposits underlie parts of France, and steep cliffs are often seen where they meet the sea in places such as the Dover cliffs on the Kent coast of the English Channel.
Chalk is mined for use in industry, such as for quicklime, bricks and builder's putty, and in agriculture, for raising pH in soils with high acidity. It is also used for "blackboard chalk" for writing and drawing on various types of surfaces, although these can also be manufactured from other carbonate-based minerals, or gypsum. (Full article...) - Image 12

Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum.
Rutile derives its name from the Latin rutilus ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner using specimens obtained in Horcajuelo de la Sierra, Madrid (Spain), which is consequently the type locality. (Full article...) - Image 13

Chalcopyrite (/ˌkælkəˈpaɪˌraɪt, -koʊ-/ KAL-kə-PY-ryte, -koh-) is a copper iron sulfide mineral and the most abundant copper ore mineral. It has the chemical formula CuFeS2 and crystallizes in the tetragonal system. It has a brassy to golden yellow color and a hardness of 3.5 to 4 on the Mohs scale. Its streak is diagnostic as green-tinged black.
On exposure to air, chalcopyrite tarnishes to a variety of oxides, hydroxides, and sulfates. Associated copper minerals include the sulfides bornite (Cu5FeS4), chalcocite (Cu2S), covellite (CuS), digenite (Cu9S5); carbonates such as malachite and azurite, and rarely oxides such as cuprite (Cu2O). It is rarely found in association with native copper. Chalcopyrite is a conductor of electricity.
Copper can be extracted from chalcopyrite ore using various methods. The two predominant methods are pyrometallurgy and hydrometallurgy, the former being the most commercially viable. (Full article...) - Image 14

Micas (/ˈmaɪkəz/ MY-kəz) are a group of silicate minerals whose outstanding physical characteristic is that individual mica crystals can easily be split into extremely thin elastic plates. This characteristic is described as perfect basal cleavage. Mica is common in igneous and metamorphic rock and is occasionally found as small flakes in sedimentary rock. It is particularly prominent in many granites, pegmatites, and schists, and "books" (large individual crystals) of mica several feet across have been found in some pegmatites.
Micas are used in products such as drywalls, paints, fillers, especially in parts for automobiles, roofing and shingles, as well as in electronics. The mineral is used in cosmetics and food to add "shimmer" or "frost." (Full article...) - Image 15

Borax (also referred to as sodium borate, tincal (/ˈtɪŋkəl/) and tincar (/ˈtɪŋkər/)) is a salt (ionic compound), a hydrated or anhydrous borate of sodium, with the chemical formula Na2H20B4O17 (also written as Na2B4O7·10H2O).
It is a colorless crystalline solid that dissolves in water to make a basic solution.
It is commonly available in powder or granular form and has many industrial and household uses, including as a pesticide, as a metal soldering flux, as a component of glass, enamel, and pottery glazes, for tanning of skins and hides, for artificial aging of wood, as a preservative against wood fungus, and as a pharmaceutic alkalizer. In chemical laboratories, it is used as a buffering agent.
The terms tincal and tincar refer to native borax, historically mined from dry lake beds in various parts of Asia. (Full article...) - Image 16
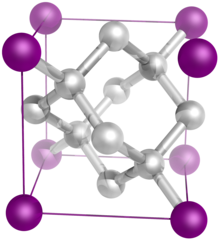
The diamond crystal structure belongs to the face-centered cubic lattice, with a repeated two-atom pattern.
In crystallography, a crystal system is a set of point groups (a group of geometric symmetries with at least one fixed point). A lattice system is a set of Bravais lattices. Space groups are classified into crystal systems according to their point groups, and into lattice systems according to their Bravais lattices. Crystal systems that have space groups assigned to a common lattice system are combined into a crystal family.
The seven crystal systems are triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic. Informally, two crystals are in the same crystal system if they have similar symmetries (though there are many exceptions). (Full article...) - Image 17
Asbestos (/æsˈbɛstəs, æz-, -tɒs/ ass-BES-təs, az-, -toss) is a naturally occurring fibrous silicate mineral. There are six types, all of which are composed of long and thin fibrous crystals, each fibre (particulate with length substantially greater than width) being composed of many microscopic "fibrils" that can be released into the atmosphere by abrasion and other processes. Inhalation of asbestos fibres can lead to various dangerous lung conditions, including mesothelioma, asbestosis, and lung cancer. As a result of these health effects, asbestos is considered a serious health and safety hazard.
Archaeological studies have found evidence of asbestos being used as far back as the Stone Age to strengthen ceramic pots, but large-scale mining began at the end of the 19th century when manufacturers and builders began using asbestos for its desirable physical properties. Asbestos is an excellent thermal and electrical insulator, and is highly fire resistant, so for much of the 20th century, it was very commonly used across the world as a building material (particularly for its fire-retardant properties), until its adverse effects on human health were more widely recognized and acknowledged in the 1970s. Many buildings constructed before the 1980s contain asbestos.
The use of asbestos for construction and fireproofing has been made illegal in many countries. Despite this, at least 100,000 people are thought to die each year from diseases related to asbestos exposure.[citation needed] In part, this is because many older buildings still contain asbestos; in addition, the consequences of exposure can take decades to arise. The latency period (from exposure to the diagnosis of negative health effects) is typically 20 years. The most common diseases associated with chronic asbestos exposure are asbestosis (scarring of the lungs due to asbestos inhalation) and mesothelioma (a type of cancer).
Many developing countries still support the use of asbestos as a building material, and mining of asbestos is ongoing, with the top producer, Russia, having an estimated production of 790,000 tonnes in 2020. (Full article...) - Image 18

Cinnabar (/ˈsɪnəˌbɑːr/; from Ancient Greek κιννάβαρι (kinnábari)), or cinnabarite (/ˌsɪnəˈbɑːraɪt/), also known as mercurblende is the bright scarlet to brick-red form of mercury(II) sulfide (HgS). It is the most common source ore for refining elemental mercury and is the historic source for the brilliant red or scarlet pigment termed vermilion and associated red mercury pigments.
Cinnabar generally occurs as a vein-filling mineral associated with volcanic activity and alkaline hot springs. The mineral resembles quartz in symmetry and it exhibits birefringence. Cinnabar has a mean refractive index near 3.2, a hardness between 2.0 and 2.5, and a specific gravity of approximately 8.1. The color and properties derive from a structure that is a hexagonal crystalline lattice belonging to the trigonal crystal system, crystals that sometimes exhibit twinning.
Cinnabar has been used for its color since antiquity in the Near East, including as a rouge-type cosmetic, in the New World since the Olmec culture, and in China since as early as the Yangshao culture, where it was used in coloring stoneware.
Associated modern precautions for the use and handling of cinnabar arise from the toxicity of the mercury component, which was recognized as early as ancient Rome. (Full article...) - Image 19

Talc, or talcum, is a clay mineral composed of hydrated magnesium silicate, with the chemical formula Mg3Si4O10(OH)2. Talc in powdered form, often combined with corn starch, is used as baby powder. This mineral is used as a thickening agent and lubricant. It is an ingredient in ceramics, paints, and roofing material. It is a main ingredient in many cosmetics. It occurs as foliated to fibrous masses, and in an exceptionally rare crystal form. It has a perfect basal cleavage and an uneven flat fracture, and it is foliated with a two-dimensional platy form.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 1 as the hardness of talc, the softest mineral. When scraped on a streak plate, talc produces a white streak, though this indicator is of little importance, because most silicate minerals produce a white streak. Talc is translucent to opaque, with colors ranging from whitish grey to green with a vitreous and pearly luster. Talc is not soluble in water, and is slightly soluble in dilute mineral acids.
Soapstone is a metamorphic rock composed predominantly of talc. (Full article...) - Image 20A sample of andesite (dark groundmass) with amygdaloidal vesicles filled with zeolite. Diameter of view is 8 cm.
Andesite (/ˈændəzaɪt/) is a volcanic rock of intermediate composition. In a general sense, it is the intermediate type between silica-poor basalt and silica-rich rhyolite. It is fine-grained (aphanitic) to porphyritic in texture, and is composed predominantly of sodium-rich plagioclase plus pyroxene or hornblende.
Andesite is the extrusive equivalent of plutonic diorite. Characteristic of subduction zones, andesite represents the dominant rock type in island arcs. The average composition of the continental crust is andesitic. Along with basalts, andesites are a component of the Martian crust.
The name andesite is derived from the Andes mountain range, where this rock type is found in abundance. It was first applied by Christian Leopold von Buch in 1826. (Full article...) - Image 21The 423-carat (85 g) blue Logan Sapphire
Sapphire is a precious gemstone, a variety of the mineral corundum, consisting of aluminium oxide (α-Al2O3) with trace amounts of elements such as iron, titanium, cobalt, lead, chromium, vanadium, magnesium, boron, and silicon. The name sapphire is derived from the Latin word sapphirus, itself from the Greek word sappheiros (σάπφειρος), which referred to lapis lazuli. It is typically blue, but natural "fancy" sapphires also occur in yellow, purple, orange, and green colors; "parti sapphires" show two or more colors. Red corundum stones also occur, but are called rubies rather than sapphires. Pink-colored corundum may be classified either as ruby or sapphire depending on locale. Commonly, natural sapphires are cut and polished into gemstones and worn in jewelry. They also may be created synthetically in laboratories for industrial or decorative purposes in large crystal boules. Because of the remarkable hardness of sapphires – 9 on the Mohs scale (the third hardest mineral, after diamond at 10 and moissanite at 9.5) – sapphires are also used in some non-ornamental applications, such as infrared optical components, high-durability windows, wristwatch crystals and movement bearings, and very thin electronic wafers, which are used as the insulating substrates of special-purpose solid-state electronics such as integrated circuits and GaN-based blue LEDs. Sapphire is the birthstone for September and the gem of the 45th anniversary. A sapphire jubilee occurs after 65 years. (Full article...) - Image 22A ruby crystal from Dodoma Region, Tanzania
Ruby is a pinkish red to blood-red colored gemstone, a variety of the mineral corundum (aluminium oxide). Ruby is one of the most popular traditional jewelry gems and is very durable. Other varieties of gem-quality corundum are called sapphires. Ruby is one of the traditional cardinal gems, alongside amethyst, sapphire, emerald, and diamond. The word ruby comes from ruber, Latin for red. The color of a ruby is due to the element chromium.
Some gemstones that are popularly or historically called rubies, such as the Black Prince's Ruby in the British Imperial State Crown, are actually spinels. These were once known as "Balas rubies".
The quality of a ruby is determined by its color, cut, and clarity, which, along with carat weight, affect its value. The brightest and most valuable shade of red, called blood-red or pigeon blood, commands a large premium over other rubies of similar quality. After color follows clarity: similar to diamonds, a clear stone will command a premium, but a ruby without any needle-like rutile inclusions may indicate that the stone has been treated. Ruby is the traditional birthstone for July and is usually pinker than garnet, although some rhodolite garnets have a similar pinkish hue to most rubies. The world's most valuable ruby to be sold at auction is the Sunrise Ruby. (Full article...) - Image 23Halite from the Wieliczka salt mine, Małopolskie, Poland
Halite (/ˈhælaɪt, ˈheɪlaɪt/ HAL-yte, HAY-lyte), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride (NaCl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, pink, red, orange, yellow or gray depending on inclusion of other materials, impurities, and structural or isotopic abnormalities in the crystals. It commonly occurs with other evaporite deposit minerals such as several of the sulfates, halides, and borates. The name halite is derived from the Ancient Greek word for "salt", ἅλς (háls). (Full article...) - Image 24

Turquoise is an opaque, blue-to-green mineral that is a hydrous phosphate of copper and aluminium, with the chemical formula CuAl6(PO4)4(OH)8·4H2O. It is rare and valuable in finer grades and has been prized as a gemstone for millennia due to its hue.
Like most other opaque gems, turquoise has been devalued by the introduction of treatments, imitations, and synthetics into the market. The robin egg blue or sky blue color of the Persian turquoise mined near the modern city of Nishapur, Iran, has been used as a guiding reference for evaluating turquoise quality. (Full article...) - Image 25
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism.
Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks.
The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ferrous-ferric oxide. (Full article...)
Selected mineralogist
- Image 1

Otto Wilhelm Hermann Abich (11 December 1806 – 1 July 1886) was a German mineralogist and geologist who was among the first to conduct scientific studies in the Caucasus region and has been called the Father of Caucasian Geology. He worked briefly at the University of Dorpat and then travelled as part of the Corps of Mining Engineers of the Russian Empire. He collected minerals and fossils across the regions in which he travelled including Armenia, Persia, Italy and Daghestan. (Full article...) - Image 2

Petru Poni (4 January 1841 – 2 April 1925) was a Moldavian (later Romanian) chemist and mineralogist.
Born into a family of răzeși (free peasants) in Săcărești, Iași County, he attended primary school in Târgu Frumos. In 1852, he enrolled in Academia Mihăileană; among his teachers were August Treboniu Laurian and Simion Bărnuțiu. He entered the University of Paris in 1865, studying chemistry there. He returned home following graduation, teaching physics and chemistry at Iași's National College and at the military high school. In 1878, he became a professor at the University of Iași, at first teaching at the medicine and science faculties, later only in the mineral chemistry department of the latter. He served as Religious Affairs and Education Minister three times: in 1891, 1895–1896 and in 1918. A bitter rival of his was the Conservative Titu Maiorescu, and he was obliged to leave the Liberal cabinet in 1896 after a dispute related to the Romanian Orthodox Church. When not in government, he continued to work in his chemistry laboratory in Iași. (Full article...) - Image 3Friedrich Klockmann (12 April 1858, Schwerin – 17 November 1937, Aachen) was a German geologist and mineralogist.
He studied geology and mineralogy at the Bergakademie in Clausthal and at the University of Rostock, receiving his doctorate in 1881. Following graduation he worked for the Prussian Geological Survey, based in Berlin. From 1887 he taught classes in mineralogy and geology at the academy in Clausthal, and in 1892 attained the title of professor. From 1899 he worked as a professor at the technical university in Aachen, where in 1917/18 he served as academic rector. (Full article...) - Image 4Stanley Hay Umphray Bowie FRS (born 24 March 1917, in Bixter, Shetland - died 3 September 2008) was a Scottish geologist. He was considered a "world authority on uranium geology and a leader in the field of geochemistry and mineralogy". He developed methods and tools to identify opaque minerals using micro-indentation hardness and optical reflectance. He worked for the British Geological Survey between 1946 and 1977. The mineral bowieite was so named in recognition of his work on identification of opaque minerals. (Full article...)
- Image 5Karl Hugo Strunz (24 February 1910 – 19 April 2006) was a German mineralogist. He is best known for creating the Nickel-Strunz classification, the ninth edition of which was published together with Ernest Henry Nickel. (Full article...)
- Image 6Lewis Caleb Beck (4 October 1798 Schenectady – 20 April 1853 Albany, New York) was an American physician, botanist, chemist, and mineralogist. The standard author abbreviation L.C.Beck is used to indicate this person as the author when citing a botanical name.[1] (Full article...)
- Image 7

Matthias Joseph Anker.
Matthias Joseph Anker (6 May 1771 – 3 April 1843) was an Austrian mineralogist and geologist born in Graz. Some sources place his birthdate as 1 May 1772.
He received his education in Vienna, afterwards working as a surgeon in the town of Stainz. In 1807, he was called to Graz as a district surgeon, from where he intensified his scientific studies in mineralogy. Four years later, he joined the staff of mineralogist Friedrich Mohs at the Johanneum in Graz, where he eventually became a professor of mineralogy as well as curator of the mineral cabinet. In 1839, he resigned from his teaching position, but stayed on as director of collections. (Full article...) - Image 8Johann Gottlob von Kurr (15 January 1798, Sulzbach an der Murr – 9 May 1870, Stuttgart) was a German pharmacist and naturalist, making contributions in the fields of botany and mineralogy.
He worked for several years as a pharmacist in Calw and other communities, then later studied medicine and surgery at the University of Tübingen, where in 1832 he received doctorates for both disciplines. From 1832 to 1870 he taught classes in natural history at the vocational school in Stuttgart (in 1841 it became known as a polytechnic institute). He was a member of the Vereins für vaterländische Naturkunde in Württemberg (Association for Natural History in Württemberg), and from 1844, was curator of its geognostic-paleontological collections. (Full article...) - Image 9
Nineteenth century drawing of Johann Friedrich Christian Hessel
Johann Friedrich Christian Hessel (27 April 1796 – 3 June 1872) was a German physician (MD, University of Würzburg, 1817) and professor of mineralogy (PhD, University of Heidelberg, 1821) at the University of Marburg. (Full article...) - Image 10Ralph Early Grim (February 25, 1902 – August 19, 1989) was an American geologist and scientist, often referred to as the "Father of Mineralogy" because he made many discoveries during his investigations of clay materials. He was one of the most outstanding mineralogists of his time and was well-known throughout the world in the field of clay science and technology. Grim's career spanned over 60 years and he received many honors and awards in the field of mineralogy. Some of the textbooks he wrote have been standard university textbooks in mineralogy for many years. (Full article...)
- Image 11

Joan Abella
Joan Abella i Creus (Sabadell, Barcelona, 1968) is a Catalan gemmologist and mineralogist who discovered abellaite, a mineral that receives this name in his honor (Full article...) - Image 12Harald Bjørlykke (14 September 1901 – 28 February 1968) was a Norwegian geologist. (Full article...)
- Image 13Portrait by Oscar Pereira da Silva
José Bonifácio de Andrada e Silva (Portuguese pronunciation: [ʒuˈzɛ boniˈfasju dʒi ɐ̃ˈdɾadɐ i ˈsiwvɐ]; 13 June 1763 – 6 April 1838) was a Brazilian statesman, naturalist, mineralist, professor and poet, born in Santos, São Paulo, then part of the Portuguese Empire.
He was one of the most important mentors of Brazilian independence, and his actions were decisive for the success of Emperor Pedro I. He supported public education, was an abolitionist and suggested that a new national capital be created in Brazil's underdeveloped interior (effected over a century later as Brasília). His career as naturalist was marked by the discovery of four new minerals. (Full article...) - Image 14

James R. Gregory, circa 1880
James Reynolds Gregory (29 December 1832 – 15 December 1899) was a noted 19th-century British mineralogist. He founded a mineral specimen business in 1858 which is today known as Gregory, Bottley & Lloyd. Gregory's company had a reputation as one of the best in the business providing mineral samples for scientists as well as private collectors. He primarily bought his specimens at auction or from other collectors and dealers, rarely collecting from the field.
When he was sent in 1868 by diamond merchant Harry Emmanuel of London's Hatton Garden to Hopetown, South Africa, to determine if claims of diamonds being found there were true, he investigated and reported back, that "The whole story of the Cape diamond discoveries is false, and is simply one of the many schemes for trying to promote the employment and expenditure of capital in searching for this pereachous [sic] substance in the colony". (Full article...) - Image 15
Alexander von Schrenk
Alexander Gustav von Schrenk (4 February 1816 – 25 June 1876) was a Baltic German-Russian naturalist born near Tula in what was then the Russian Empire. He was a brother to zoologist Leopold von Schrenck (1826–1894).
From 1834 to 1837, he studied sciences at the University of Dorpat (Tartu), later spending several years as an assistant at the botanical garden in St. Petersburg. He was habilitated for mineralogy at Dorpat, where from 1849 he served as a lecturer. From 1858 he spent the next ten years at his wife's manor in Pühajärve (Heiligensee), Livonia, returning to Dorpat in 1868, where he died several years later. (Full article...) - Image 16Arthur Hutchinson by Sir William Rothenstein
Arthur Hutchinson OBE FRS (6 July 1866 – 12 December 1937) was a British mineralogist. During World War I, and at the request of the Admiralty, he was asked to design gas masks suitable for the Navy; for his work, he was awarded the OBE. Hutchinson was elected a Fellow of the Royal Society in 1922. He was master of Pembroke College, Cambridge, from 1928 to 1937, served on the Council of the Royal Society from 1932 to 1934, and was the Society's Vice-President for the year 1933-34. (Full article...) - Image 17Dr. E-An Zen (任以安) was born in Peking, China, May 31, 1928, and came to the U.S. in 1946. He became a citizen in 1963. Since 1990 he was adjunct professor at the University of Maryland. He died on March 29, 2014, at the age of 85.
He has contributed articles to professional journals and is a fellow of the Geological Society of America (Councillor, 1985–88, 1990–93; President, 1991–92); the American Association for the Advancement of Science (AAAS), the American Academy of Arts and Sciences, the Mineralogical Society of America (Council, 1974–77;Pres., 1975–76). He is a member of the Geological Society of Washington (Pres. 1973), the National Academy of Sciences, and the Mineralogical Association of Canada. Zen has been active in programs to bring geological knowledge to the general public. (Full article...) - Image 18Serve-Dieu Abailard "Armand" Lévy (14 November 1795 – 29 July 1841) was a French mathematician and mineralogist. He is remembered in the Haüy-Lévy notation for describing mineral crystal structures. (Full article...)
- Image 19Andrew Ketcham Barnett (1852–1914) was a mineral collector and dealer in Penzance, Cornwall, in the United Kingdom. He was Principal of the Penzance School of Mines (now part of the Camborne School of Mines), lectured on mining, and helped to build their mineral collection. He also served as Mayor of Penzance on seven occasions from 1906 to 1913.
Cooper writes of him:
:"Barnett was born in Chacewater, Cornwall in 1852. He was an original member of the Mineralogical Society, a Fellow of the Geological Society from 1875, and President of the Royal Geological Society of Cornwall 1907–08, having been awarded their Bolitho Medal in 1906. His classes on mineralogy in 1873 led to the establishment of the Mining and Science School at Penzance of which he became Principal. He was mayor of Penzance seven times from 1906 to 1913. Active as a mineral dealer from at least 1876 to 1887 at Chyandour, Penzance, he specialized in local specimens and occasionally sold minerals brought back from Australia by "Cousin Jack." In the 1881 Census he described himself as an assayer. He is the author of the now-rare Elementary Inorganic Chemistry; lecture notes, ca. 1900." (Full article...) - Image 20

Ludwig Meyn. He was called Dr. Weisheit (Dr. Wisdom) by his friends.
Ludwig Meyn (1 October 1820, Pinneberg − 4 November 1878, Uetersen), was a German agricultural scientist, soil scientist, geologist, journalist and mineralogist. He was the pioneer of oil production. (Full article...) - Image 21

Antonio D'Achiardi (28 November 1839, in Pisa – 10 December 1902, in Pisa) was an Italian geologist and mineralogist known for his mineralogical studies of Tuscany. He was the father of the mineralogist Giovanni D'Achiardi [it], and the artist, Pietro D'Achiardi.
In 1859 he received his doctorate in sciences from the University of Pisa, afterwards working as an assistant for chemistry (from 1861). Three months after this appointment, he lost the use of his left eye due to a laboratory accident involving nitric acid. He subsequently abandoned his career in chemistry, and instead devoted his attention to geology and mineralogy, becoming a student of Giuseppe Meneghini. He later became a professor of geology at Pavia and in 1874 was appointed a professor of mineralogy at the University of Pisa. In 1881 he established a laboratory of mineralogy at Pisa. (Full article...) - Image 22

Johann Friedrich Ludwig Hausmann
Johann Friedrich Ludwig Hausmann (22 February 1782, Hannover – 26 December 1859, Göttingen) was a German mineralogist. (Full article...) - Image 23
John Walker FRSE (1731–1803) was a Scottish minister and natural historian. He was Regius Professor of Natural history at the University of Edinburgh from 1779 to 1803. He was joint founder of the Royal Society of Edinburgh in 1783 and Moderator of the General Assembly of the Church of Scotland in 1790. (Full article...) - Image 24

Henry Carvill Lewis
Henry Carvill Lewis (November 16, 1853 – July 21, 1888) was an American geologist and mineralogist. (Full article...) - Image 25
Nikolai Semyonovich Kurnakov (Russian: Николай Семёнович Курнаков; 6 December [O.S. 24 November] 1860 – 19 March 1941) was a Russian chemist, who is internationally recognized as the originator of physicochemical analysis. He also was one of the principal founders of the platinum industry in the Soviet Union. A chemical reaction that he pioneered, known as the Kurnakov test, is still used to differentiate cis from trans isomers of divalent platinum and is his best-known contribution to coordination chemistry. (Full article...)
Related portals
Get involved
For editor resources and to collaborate with other editors on improving Wikipedia's Minerals-related articles, see WikiProject Rocks and minerals.
General images
- Image 1Hübnerite, the manganese-rich end-member of the wolframite series, with minor quartz in the background (from Mineral)
- Image 4Diamond is the hardest natural material, and has a Mohs hardness of 10. (from Mineral)
- Image 6Gypsum desert rose (from Mineral)
- Image 7Sphalerite crystal partially encased in calcite from the Devonian Milwaukee Formation of Wisconsin (from Mineral)
- Image 8Mohs hardness kit, containing one specimen of each mineral on the ten-point hardness scale (from Mohs scale)
- Image 9Mohs Scale versus Absolute Hardness (from Mineral)
- Image 10Native gold. Rare specimen of stout crystals growing off of a central stalk, size 3.7 x 1.1 x 0.4 cm, from Venezuela. (from Mineral)
- Image 14Red cinnabar (HgS), a mercury ore, on dolomite. (from Mineral)
- Image 15Black andradite, an end-member of the orthosilicate garnet group. (from Mineral)
- Image 17Pink cubic halite (NaCl; halide class) crystals on a nahcolite matrix (NaHCO3; a carbonate, and mineral form of sodium bicarbonate, used as baking soda). (from Mineral)
- Image 18Perfect basal cleavage as seen in biotite (black), and good cleavage seen in the matrix (pink orthoclase). (from Mineral)
- Image 20Muscovite, a mineral species in the mica group, within the phyllosilicate subclass (from Mineral)
- Image 21An example of elbaite, a species of tourmaline, with distinctive colour banding. (from Mineral)
- Image 22Asbestiform tremolite, part of the amphibole group in the inosilicate subclass (from Mineral)
- Image 23Epidote often has a distinctive pistachio-green colour. (from Mineral)
- Image 24When minerals react, the products will sometimes assume the shape of the reagent; the product mineral is termed a pseudomorph of (or after) the reagent. Illustrated here is a pseudomorph of kaolinite after orthoclase. Here, the pseudomorph preserved the Carlsbad twinning common in orthoclase. (from Mineral)
- Image 25Schist is a metamorphic rock characterized by an abundance of platy minerals. In this example, the rock has prominent sillimanite porphyroblasts as large as 3 cm (1.2 in). (from Mineral)
In the news
- 3 May 2024 –
- Panama bans First Quantum Minerals from extracting copper following the closure of its Cobre Panamá mine last year. (Reuters) (The Globe and Mail)
Did you know ...?
- ... that the Lewisian gneiss (pictured), forms the basement to Torridonian and Cambro-Ordovician sedimentary rocks in the Hebridean Terrane?
- ... that abernathyite is both fluorescent and radioactive and is named for the mine operator who discovered it?
- ... that the minerals armalcolite, pyroxferroite and tranquillityite were discovered in lunar rocks?
- ...that the streak of a mineral, the color of the mark it makes when rubbed on a plate, is usually a more consistent identifier than the color of the original mineral?
Subcategories
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus