
Potassium ferrocyanide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Potassium ferrocyanide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with potassium ferricyanide.
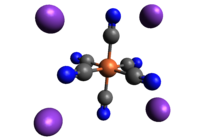
Potassium ferrocyanide is the inorganic compound with formula K4[Fe(CN)6]·3H2O. It is the potassium salt of the coordination complex [Fe(CN)6]4−. This salt forms lemon-yellow monoclinic crystals.
Quick Facts Names, Identifiers ...
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Potassium hexacyanidoferrate(II) | |
| Other names | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.034.279 |
| EC Number |
|
| E number | E536 (acidity regulators, ...) |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| K4[Fe(CN)6] | |
| Molar mass | 368.35 g/mol (anhydrous) 422.388 g/mol (trihydrate) |
| Appearance | Light yellow, crystalline granules |
| Density | 1.85 g/cm3 (trihydrate) |
| Boiling point | (decomposes) |
| trihydrate 28.9 g/100 mL (20 °C) | |
| Solubility | insoluble in ethanol, ether |
| −130.0·10−6 cm3/mol | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H411 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
6400 mg/kg (oral, rat)[3] |
| Related compounds | |
Other anions |
Potassium ferricyanide |
Other cations |
Sodium ferrocyanide Prussian blue |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
