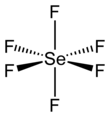
Selenium hexafluoride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Selenium hexafluoride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Selenium hexafluoride is the inorganic compound with the formula SeF6. It is a very toxic colourless gas described as having a "repulsive" odor.[5] It is not widely encountered and has no commercial applications.[6]
Quick Facts Names, Identifiers ...
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Selenium hexafluoride | |||
| Other names
Selenium(VI) fluoride, Selenium fluoride | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider |
| ||
| ECHA InfoCard | 100.149.506 | ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| SeF6 | |||
| Molar mass | 192.9534 g/mol | ||
| Appearance | colourless gas | ||
| Density | 0.007887 g/cm3[1] | ||
| Melting point | −39 °C (−38 °F; 234 K) | ||
| Boiling point | −34.5 °C (−30.1 °F; 238.7 K) sublimes | ||
| insoluble | |||
| Vapor pressure | >1 atm (20°C)[2] | ||
| −51.0·10−6 cm3/mol | |||
Refractive index (nD) |
1.895 | ||
| Structure | |||
| Orthorhombic, oP28 | |||
| Pnma, No. 62 | |||
| octahedral (Oh) | |||
| 0 | |||
| Thermochemistry | |||
Std enthalpy of formation (ΔfH⦵298) |
-1030 kJ/mol[3] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
toxic, corrosive | ||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published) |
10 ppm (rat, 1 hr) 10 ppm (mouse, 1 hr) 10 ppm (guinea pig, 1 hr)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 0.05 ppm (0.4 mg/m3)[2] | ||
REL (Recommended) |
TWA 0.05 ppm[2] | ||
IDLH (Immediate danger) |
2 ppm[2] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close