
Sodium naphthalene
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Sodium naphthalene?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
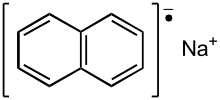
Sodium naphthalene is an organic salt with the chemical formula Na+[C10H8]−. In the research laboratory, it is used as a reductant in the synthesis of organic, organometallic, and inorganic chemistry. It is usually generated in situ. When isolated, it invariably crystallizes as a solvate with ligands bound to Na+.[1]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium naphthalenide | |
| Systematic IUPAC name
Sodium naphthalen-1-ide | |
| Other names
sodium naphthalenide, sodium naphthalide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.020.420 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Na+[C10H8]− | |
| Molar mass | 151.164 g·mol−1 |
| Appearance | Deep green crystals |
| Related compounds | |
Other anions |
Lithium naphthalene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close